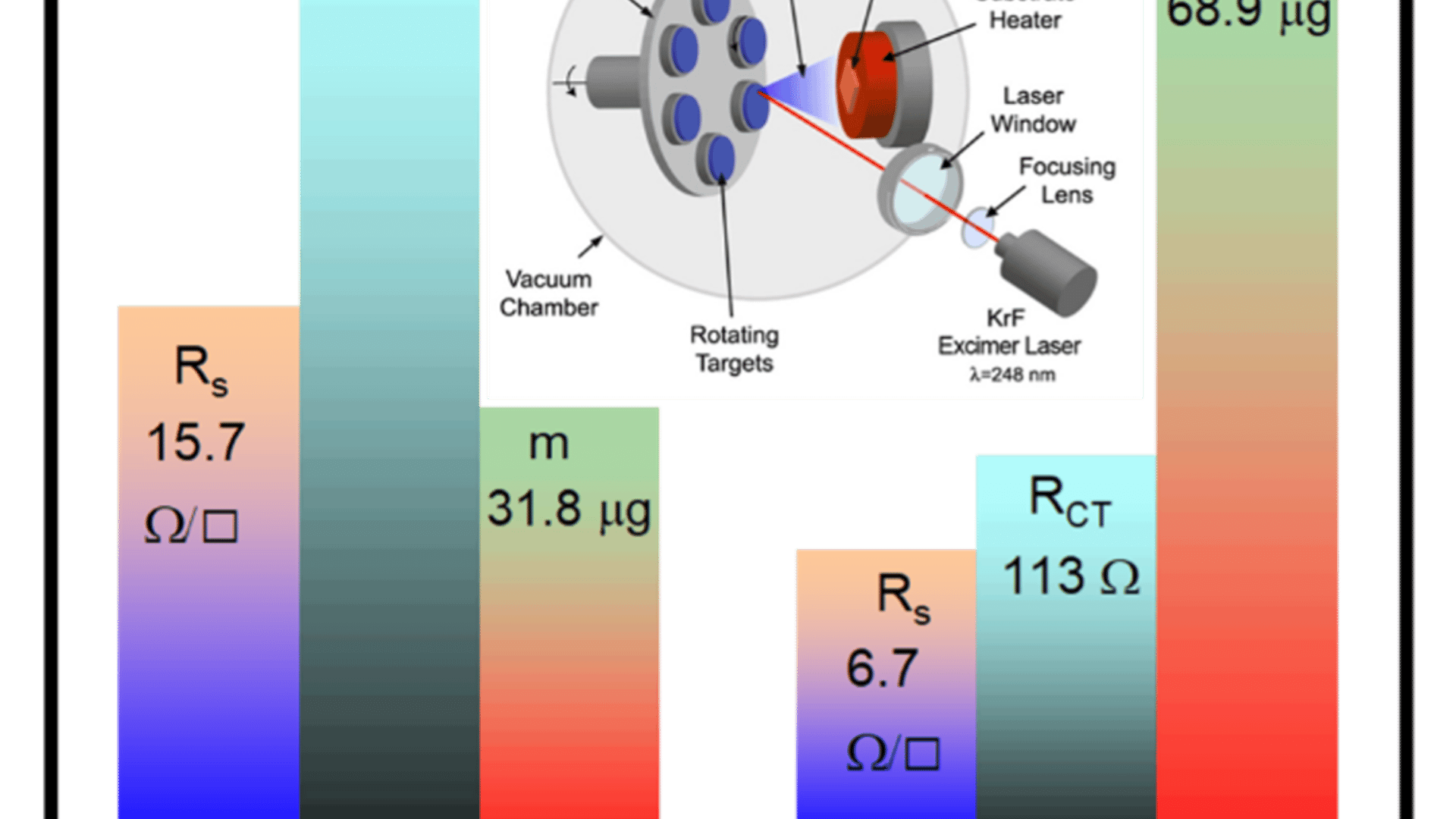
Researchers at North Carolina A&T State University have developed a laser-made thin-film catalyst that could help lower the cost of producing clean hydrogen from water. Using a process called pulsed laser deposition, the team grew ruthenium oxide films that drive the oxygen-evolution reaction — the harder half of water splitting — on sapphire, a much less expensive substrate than those typically used in this kind of thin-film research.
“The importance of high-quality ruthenium oxide thin films on sapphire substrates stems from two aspects,” says Dhananjay Kumar, mechanical engineering professor at NCAT and PI for the PREM project, Collaborative Research and Education in Advanced Materials. “The first aspect is associated with the cost of sapphire substrates, which is almost an order of magnitude lower than other substrates commonly used for the growth of films for electrocatalysis studies. The second aspect is associated with the importance of sapphire as a technologically important substrate material.” According to Kumar, who supervised this research, after silicon, there is no substrate material other than sapphire that can be produced in a single crystal large size wafer.
The team recently published their research in ACS Applied Energy Materials.
While choosing sapphire is part of the story, perhaps the bigger takeaway is that the researchers showed they could get strong performance on a lower-cost platform. Their best-performing film achieved an overpotential of 280 millivolts at 100 microamps per square centimeter, while also showing lower charge-transfer resistance and better stability than the thinner version they tested. In practical terms, that means the material needed less extra energy to keep the reaction going and held up better over time.
The study also found that thickness matters. The thicker film outperformed the thinner one largely because its lower electrical resistance allowed charges to move more easily through the material during the reaction. That finding gives researchers a clearer design rule for improving future catalysts: not just what material to use, but how to build it for better performance.
“The study has enabled us to gain an understanding of the role of the sample’s resistance in the charge transfer process between electrode surface and electrolyte — something that hasn’t been reported in the literature before,” says Kumar.